Have you ever added sugar to your tea and watched it disappear? Or sprinkled salt into water and noticed it vanish completely? That is the fascinating effect of substances that can dissolve. In simple terms, a substance that can dissolve, often in water, breaks into tiny particles, spreads evenly, and forms a clear solution.
Yet, the concept of dissolving goes beyond the kitchen. In chemistry, it explains how materials interact with liquids. In biology, it describes vitamins, minerals, and fibers that travel through the body. Even in everyday language, the term can refer to something that is solvable or fixable. Therefore, this guide provides a complete overview, including practical examples, tables, scientific explanations, and applications, all organized for easy understanding.
Simple Definition of Soluble
At its core, being able to dissolve in a liquid means a substance can mix completely to form a solution. In this process, the particles of the material spread evenly throughout the liquid.
Examples of substances that dissolve:
- Sucrose (table sugar) dissolves easily in water.
- Sodium chloride (table salt) mixes completely in water.
Examples of substances that do not dissolve:
- Sand
- Oil
Origin: The term comes from the Latin word solvere, meaning “to loosen” or “to untie,” which reflects how particles separate and disperse when mixed with a liquid.
Example sentences:
- “Salt dissolves easily in water.”
- “The powder mixes quickly at higher temperatures.”
Soluble in Chemistry
In chemistry, “soluble” is a key concept. Specifically, it describes how well a substance dissolves in a solvent. When a substance dissolves:
- Its solid breaks into tiny particles.
- These particles spread evenly through the liquid.
- Finally, a clear solution forms.
Examples:
| Substance | Solubility in Water | Notes |
|---|---|---|
| Sodium chloride | High | Dissolves completely at room temperature |
| Sucrose | Highly soluble | Forms a sweet solution |
| Calcium carbonate | Poorly soluble | Needs acid or heat to dissolve |
Explanation:
Water is polar, which allows it to pull apart ions and polar molecules. On the other hand, non-polar substances, like oil, can’t mix with water because their molecules don’t match. Therefore, understanding solubility helps predict reactions, make solutions, and separate mixtures in chemistry labs.
How Dissolving Works
Dissolving is a molecular process. Take, for instance, sodium chloride. Salt contains positively charged sodium ions and negatively charged chloride ions. When water is added:
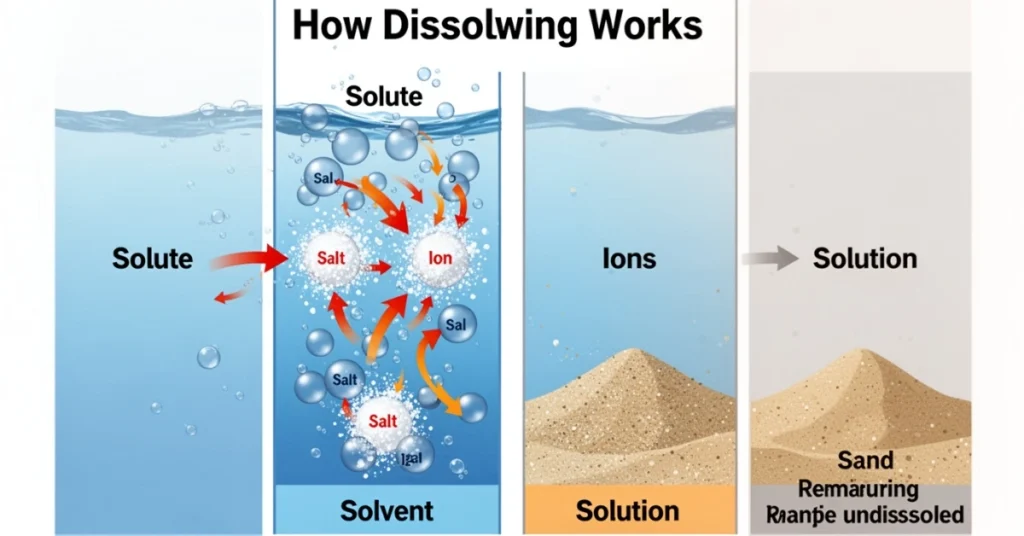
- Water molecules surround the ions.
- Then, the charges pull the ions apart.
- Finally, ions spread evenly, forming a clear solution.
- However, some substances, like sand, don’t dissolve because water molecules can’t separate their particles. Moreover, dissolving also depends on temperature, pressure, particle size, and stirring.
Everyday Examples of Soluble Substances
Understanding soluble substances becomes easier with examples. Here is a table of common soluble substances:
| Substance | Example |
|---|---|
| Sucrose | Sugar in tea, dissolved in water |
| Sodium chloride | Salt in soup, also in water |
| Vitamin C | Supplements mixed in water |
| Ethanol | Alcohol diluted in water |
In contrast, substances like sand, oil, and chalk remain separate and are insoluble.
Factors That Affect Solubility
Not all substances dissolve in the same way. Several factors influence how well a material mixes with a liquid:
- Temperature: Solids usually dissolve faster in warmer liquids, while gases tend to dissolve better in colder liquids.
- Pressure: Mainly affects gases; higher pressure allows more gas to dissolve.
- Particle Size: Smaller pieces dissolve more quickly than larger chunks.
- Stirring: Helps distribute the material evenly throughout the liquid.
- Chemical Nature: Polar substances mix well with polar liquids, while non-polar materials do not.
Example: Sugar dissolves faster in hot water than in cold water.
Soluble in Biology
In biology, soluble substances are essential for nutrient transport and metabolism.
- Vitamin C is water-soluble and moves easily through the bloodstream.
- Vitamin B12 is also water-soluble and must be replenished daily.
- Soluble fiber creates a gel in the digestive system, which helps with digestion, lowers cholesterol, and keeps blood sugar steady.
| Nutrient | Solubility | Storage |
|---|---|---|
| Vitamin C | Water-soluble | Not stored |
| Vitamin B12 | Water-soluble | Limited storage |
| Fat-soluble vitamins (A, D, E, K) | Fat-soluble | Stored in fat tissue |
Therefore, soluble nutrients must be regularly consumed for proper body function.
Soluble in Medicine and Industry
Solubility plays a crucial role in medicine and industry.
In medicine:
- Soluble drugs dissolve quickly for rapid absorption.
- Soluble fibers improve digestion and regulate cholesterol.
In industry:
- Soluble chemicals ensure uniform reactions in detergents, paints, dyes, and beverages.
- Moreover, environmental science relies on solubility to manage water-soluble pollutants.
For example, detergents rely on soluble compounds, which help them spread in water and remove dirt effectively.
Soluble in Everyday Language
Beyond science, soluble often refers to something capable of being solved.
For instance:
- “The issue is soluble with proper communication.”
Thus, soluble means the problem can be fixed, just like a substance dissolves in a liquid. This metaphor shows the versatility of the term.
Solubility of Gases and Liquids
Solubility isn’t just for solids gases and liquids can dissolve too. Carbon dioxide mixes with water to make soda fizzy. Likewise, oxygen dissolves in water to support aquatic life. However, solubility depends on temperature and pressure. Cold water dissolves gases better than warm water. Additionally, higher pressure allows more gas to dissolve.
| Gas | Cold Water Solubility | Warm Water Solubility | Example |
|---|---|---|---|
| Oxygen (O₂) | High | Low | Fish survival in lakes |
| Carbon dioxide (CO₂) | High | Low | Carbonated drinks fizz |
| Nitrogen (N₂) | Medium | Low | Aquatic ecosystems |
Practical Experiments to Observe Solubility
Hands-on experiments make solubility easy to visualize and fun:
Experiment 1: Solids in Water
- Materials: 3 cups of water, sugar, salt, sand
- Method: Add each substance to a cup, stir, observe
- Observation: Sugar and salt dissolve; sand does not
Experiment 2: Oil and Water
- Materials: 1 cup water, 1 tablespoon oil
- Method: Stir vigorously
- Observation: Oil separates, demonstrating insolubility
Tips:
- Use hot water to increase dissolving speed
- Grind solids to increase surface area
- Stir continuously for faster dissolution
Soluble vs Insoluble in Food and Nutrition
Understanding soluble and insoluble substances in food is essential for healthy digestion.
| Fiber Type | Solubility | Example Foods | Health Benefits |
|---|---|---|---|
| Soluble | Dissolves in water | Oats, beans, apples | Lowers cholesterol, regulates blood sugar |
| Insoluble | Does not dissolve | Wheat bran, cauliflower, nuts | Adds stool bulk, aids digestion |
Key Facts:
- Soluble fiber slows digestion and stabilizes blood sugar
- Insoluble fiber helps prevent constipation
- Both types are needed for a balanced diet
Industrial Applications of Solubility
Solubility has real-world industrial importance:
Bullet Points:
- Detergents: Soluble compounds ensure uniform cleaning
- Pharmaceuticals: Soluble drugs dissolve for faster absorption
- Beverage Industry: Sugar, flavorings, and additives dissolve evenly
- Environmental Science: Water-soluble pollutants are easier to monitor and control
Tips to Increase or Control Solubility
Knowing how to control solubility is useful in cooking, science, and industry.
Methods:
- Increase temperature: Sugar dissolves faster in hot tea
- Stirring: Spreads particles evenly
- Crushing or grinding solids: Increases surface area
Frequently Asked Questions (FAQs)
Q1: What does soluble mean?
A: A substance that can dissolve in a liquid
Q2: What are examples of soluble substances?
A: Sugar, salt, Vitamin C
Q3: How is soluble different from insoluble?
A: Soluble dissolves completely; insoluble does not
Q4: Is salt always soluble?
A: It dissolves readily, but temperature and pressure affect how much
Q5: What is water-soluble?
A: Substances that dissolve in water and move easily in the body
Q6: What factors affect solubility?
A: Temperature, pressure, particle size, stirring, and chemical nature
Quick Summary
In simple terms, soluble substances can dissolve in a liquid, whereas insoluble substances cannot. When a substance dissolves, it forms a solution, which is a mixture in which the particles spread evenly throughout the solvent. The solubility of a substance depends on several factors, including temperature, pressure, stirring, particle size, and chemical compatibility. In biology, water-soluble vitamins and fibers play an essential role in maintaining health, as they move easily through the body and support digestion and metabolism. Solubility is also critical in chemistry, medicine, industry, and everyday life.
Therefore, understanding solubility is key in many areas. It helps in science experiments, cooking, medicine, environmental management, and even solving everyday problems.
Click Here To Read More About : what does the name alexis mean

I am Clara Lexis, a writer driven by clarity, depth, and authenticity. My focus is on transforming ideas into meaningful content that is both informative and engaging. I write with intention to communicate clearly, thoughtfully, and with purpose.